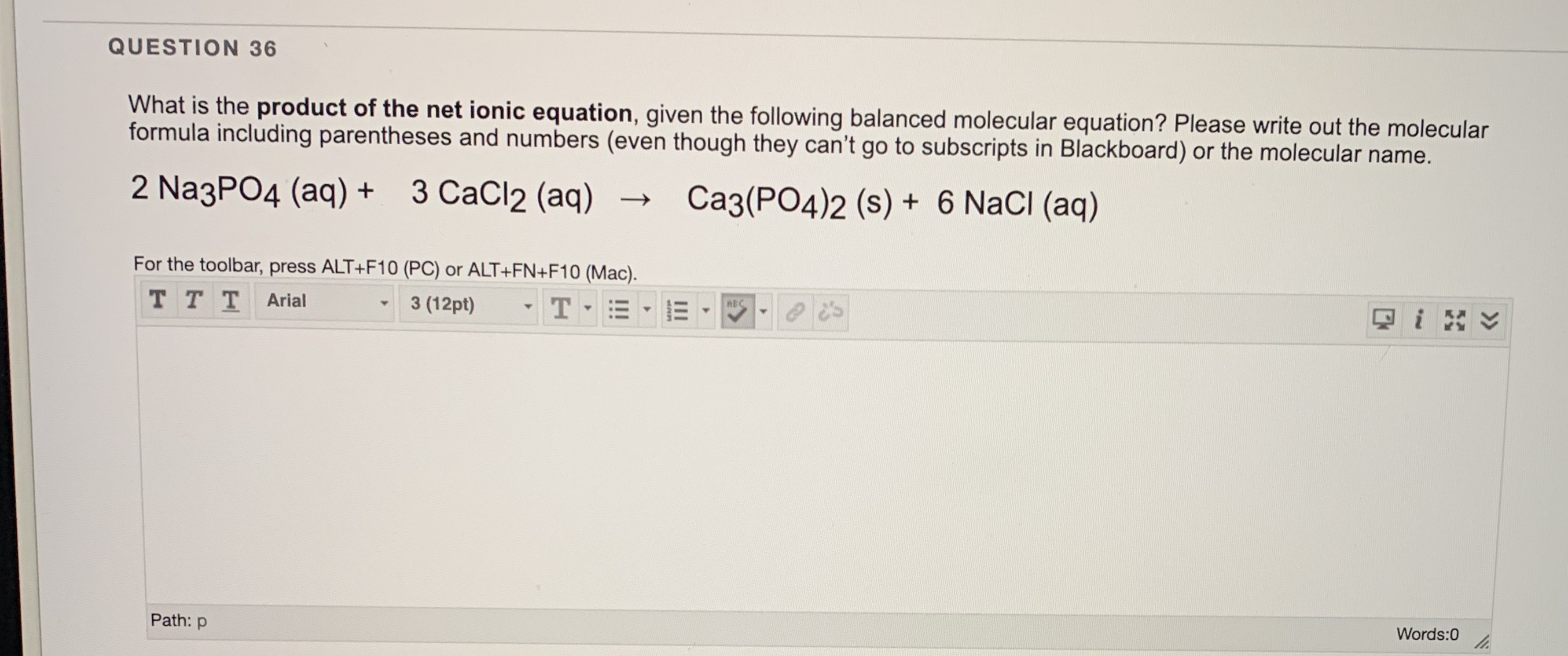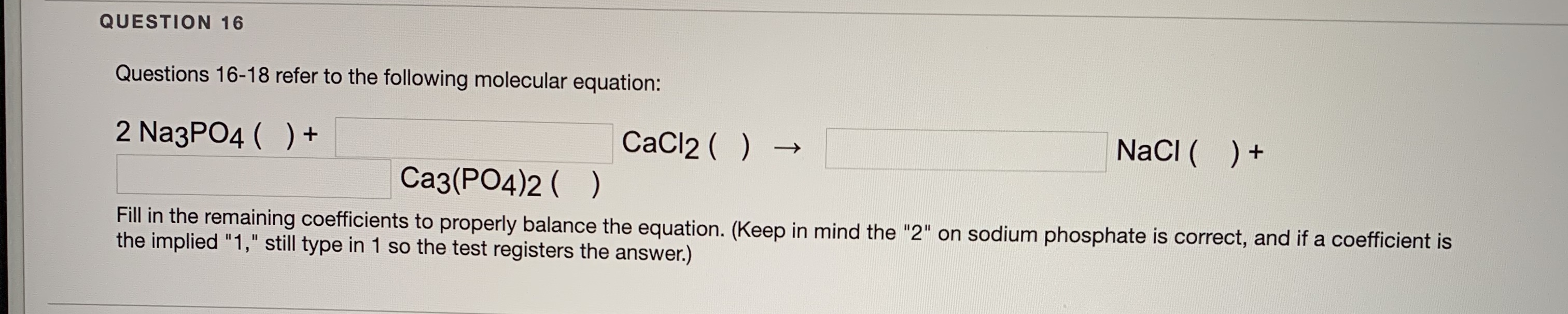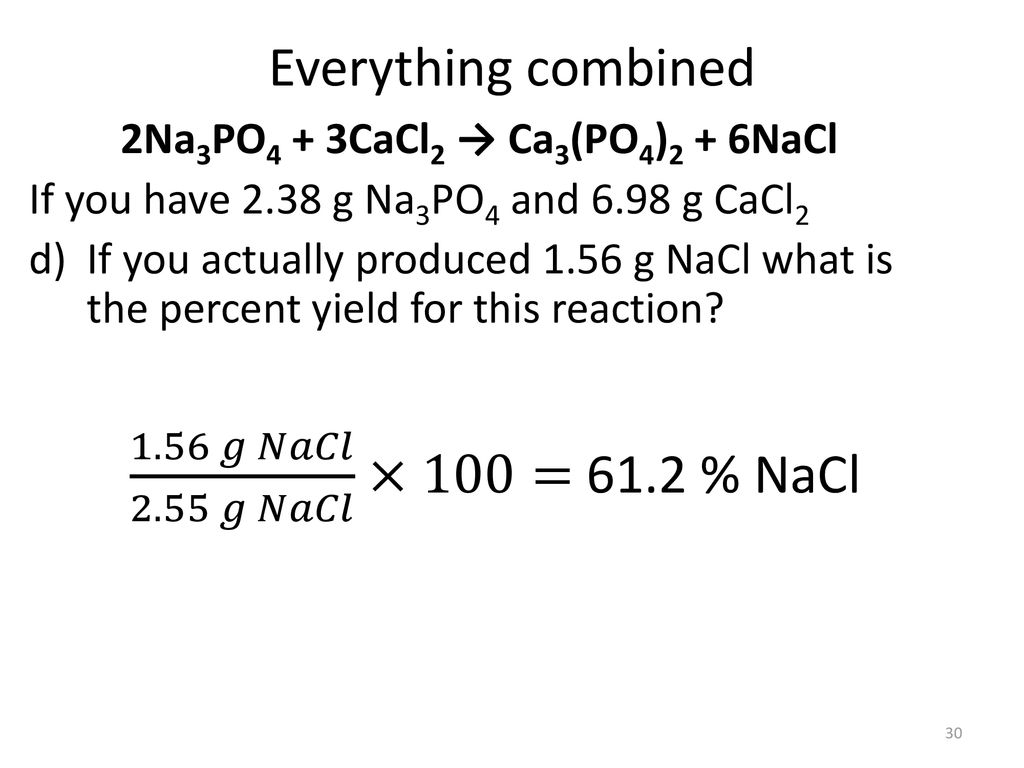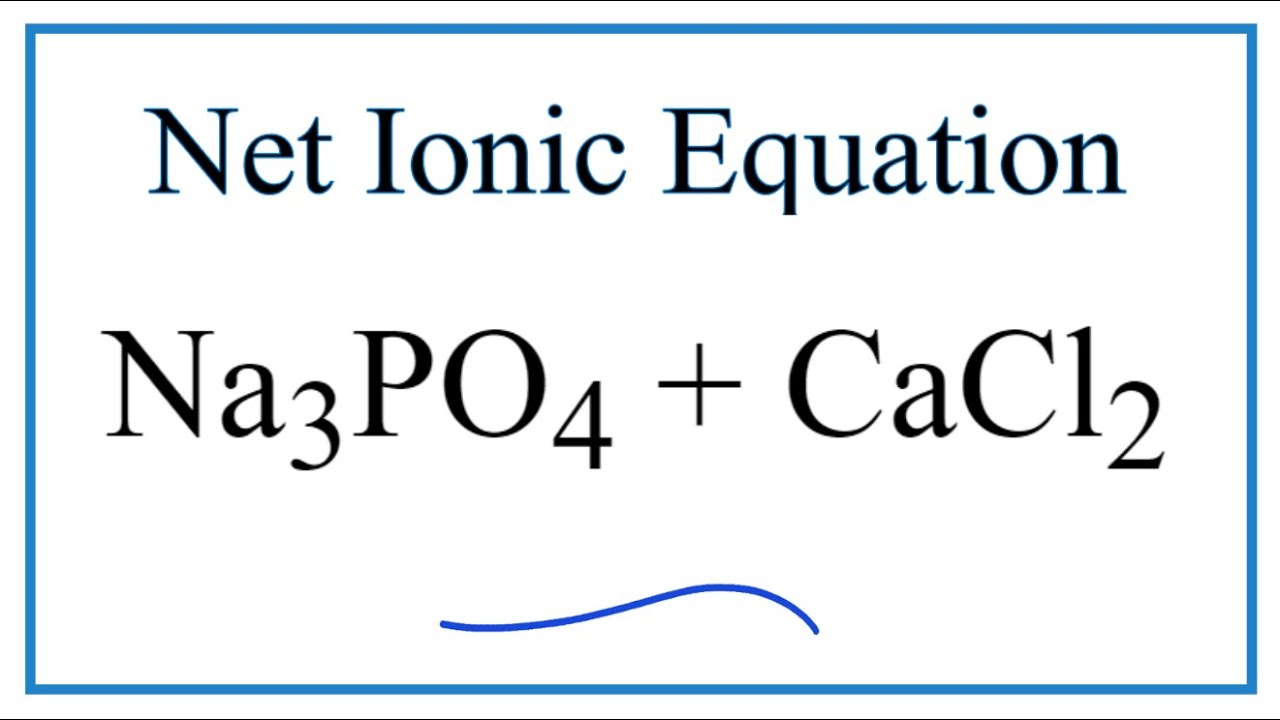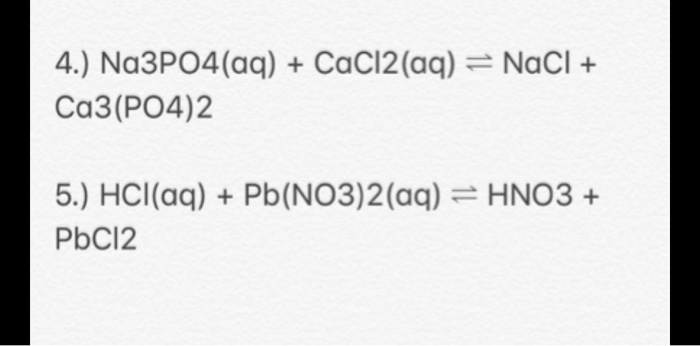
SOLVED: 10.0 g of CaCl2 and 15.0 g of Na3PO4 are combined in a double replacement reaction: CaCl2 + Na3PO4 –> Ca3(PO4)2 + NaCl a. When this equation is balanced, what is
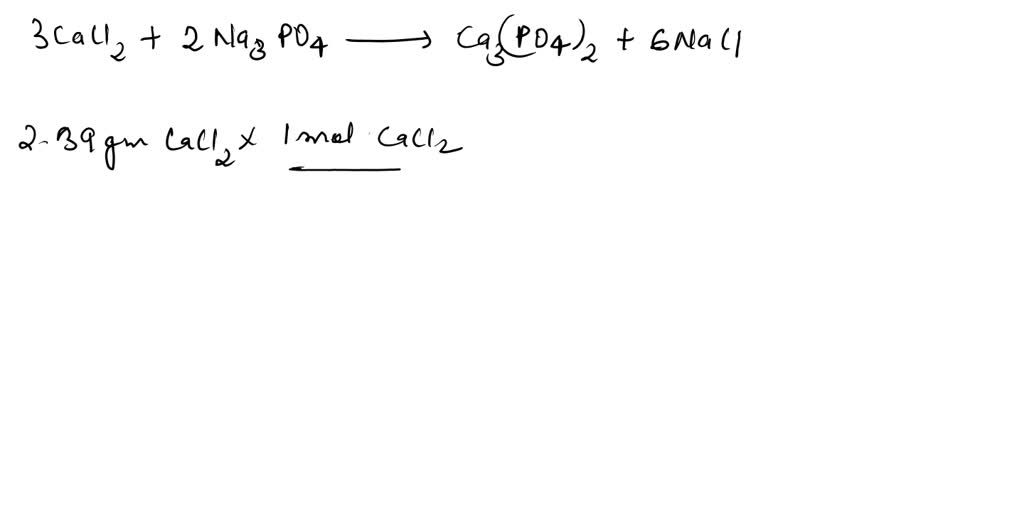
SOLVED: 3 CaCl2(aq) + 2 Na3PO4(aq) -> Ca3(PO4)2(s) + 6 NaCl(aq) 1,50 M 250,0 mL CaCl2 solution is reacted with excess Na3PO4 solution. Mass of the solid product is 36,7 g. What

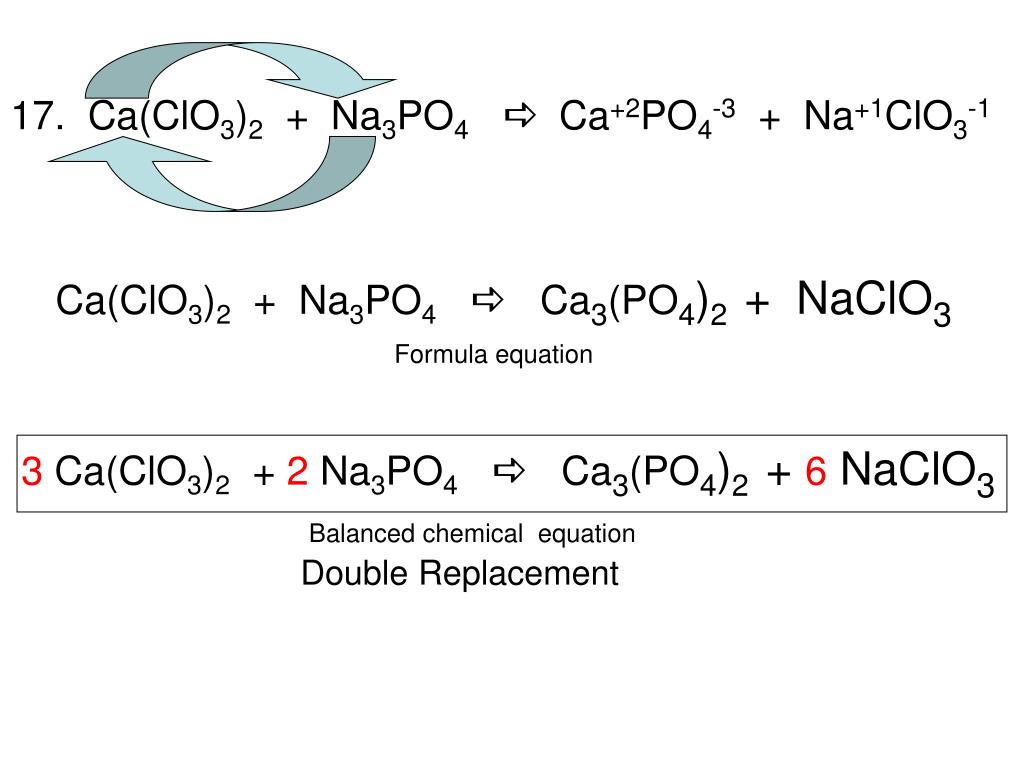
CaCl2+Na3PO4=Ca3(PO4)2 + NaCl Balanced Equation||Calcium chloride+Sodium phosphate Balanced Equation - YouTube